Quartz Beneficiation Process
Date: 2026-02-24 Categories: Non Metals Views: 17
Quartz beneficiation is a process that removes impurities from quartz ore and improves its purity through a series of physical and chemical methods. Its core process flow and key points are as follows:
I. Core Process Flow
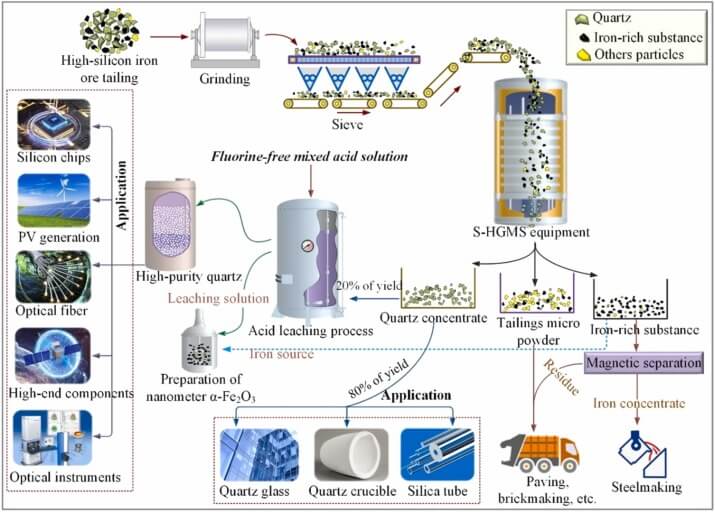
Crushing and Grinding
Purpose: To crush large quartz ore to a suitable particle size, releasing impurities and facilitating subsequent separation.
Equipment: Jaw crusher (coarse crushing), cone crusher (medium crushing), ball mill/rod mill (fine grinding).
Parameters: The final particle size is typically controlled at 0.1-0.5 mm to avoid over-grinding and introducing impurities.
Physical Separation
Magnetic Separation
Principle: Utilizing the difference in magnetic properties between quartz and iron-bearing minerals, separation is achieved through a magnetic field.
Equipment: Wet plate magnetic separator, high gradient magnetic separator (HGMS).
Effect: Removes weakly magnetic impurities such as hematite, ilmenite, and biotite.
Flotation
Principle: Adjusting the surface wettability of minerals with reagents allows quartz to separate from feldspar, mica, etc.
Reagents: Hydrofluoric acid (HF) activates the quartz surface; amine collectors (such as dodecylamine) are used for the flotation of feldspar/mica; an environmentally friendly alternative is fluorine-free flotation (organic acid + cationic collector).
Equipment: Flotation machine, flotation column.
Gravity Separation and Photoelectric Separation
Gravity Separation: Shaking tables and spiral sluices separate dense impurities (such as zircon and garnet).
Photoelectric Separation: Laser separators identify particles with abnormal color (such as red quartz containing Fe³⁺).
Deep Chemical Purification
Acid Leaching
Purpose: To dissolve metallic impurities (such as Al and Fe) on the quartz surface and in micro-cracks.
Acid Solution: Mixed acid system (HCl 6-8 mol/L + H₂SO₄ concentrate + HF 2-5%), temperature 80-100℃, time 4-8 hours.
Auxiliary Technology: Microwave/ultrasound accelerates the reaction, reducing acid usage by 30-50%.
High-Temperature Chlorination
Purpose: To remove sparingly soluble impurities such as titanium and chromium.
Conditions: Cl₂ or a mixture of Cl₂ and HCl gas is introduced at 1200-1500℃ to cause the metallic impurities to form volatile chlorides (such as TiCl₄).
Equipment: High-temperature rotary kiln or fluidized bed reactor; a tail gas treatment system is required.
Ultra-Clean Treatment and Post-Processing
Secondary Magnetic Separation: A high-gradient magnetic separator removes residual magnetic particles (such as Fe₃O₄).
Heat Treatment
Calcination: Calcination at 1400-1600℃ for 2-4 hours promotes impurity volatilization and homogenizes the quartz crystal lattice.
Water Quenching: Rapid cooling of the high-temperature quartz breaks it into small particles for easier subsequent processing.
Acid Washing (Secondary): Ultrasonic cleaning with dilute hydrochloric acid (5%) and deionized water removes surface residues.
Surface Polishing: The surface is polished with a mixture of hydrofluoric acid and nitric acid (HF: HNO₃ = 1:3) to reduce microcracks and adsorbed impurities.
Detection and Sorting
Purity Analysis: ICP-MS is used to detect metallic impurities down to the ppb level, and FTIR is used to detect hydroxyl content (<1 ppm required).
Laser Sorting: LIBS technology is used for real-time monitoring, and a robotic arm removes impurity particles.
Manual Sorting (Final Inspection): Particles with abnormal fluorescence (containing organic matter or uranium/thorium impurities) are removed under ultraviolet light.
II. Key Technologies and Challenges
Targeted Impurity Removal
Al: Relies on HF acid leaching and high-temperature chlorination (AlCl₃ sublimation point 180℃).
Ti: Requires high-temperature chlorination (TiCl₄ boiling point 136℃).
Gas-Liquid Inclusions: Ultra-high temperature (>1600℃) vacuum treatment or thermal detonation.
Environmental Protection and Safety
HF Alternatives: Such as fluorosilicic acid systems.
Cl₂ Tail Gas Recovery: Alkali spray absorption.
Wastewater Treatment: Lime neutralization of fluoride ions to generate CaF₂ precipitate.
III. Applications and Standards
Semiconductor Grade Quartz: Must meet SEMI standards (e.g., SEMI F57), hydroxyl content <1 ppm, total metallic impurities <20 ppm.
Photovoltaic Grade Quartz: Meets the requirements of GB/T 32649-2016 High-Purity Quartz Sand, Fe <8 ppm, Al <15 ppm.